When your experiment depends on accurate protein quantification, the quality of your ELISA kit is non‑negotiable. A substandard kit can lead to irreproducible results, wasted samples, and lost time—yet many researchers simply trust the manufacturer’s claims. How can you objectively verify that an ELISA test kit meets its stated specifications before or after purchase? This guide walks you through the key quality indicators and simple validation methods, empowering you to choose a reliable elisa kit supplier and ensure your data are trustworthy.
Part 1: How ELISA Kits Work
Before evaluating quality, it helps to understand the basic principle. Most modern ELISA kits use the sandwich format for cytokines and other proteins:
- Capture antibody is pre‑coated on a 96‑well plate.
- Sample or standard containing the target antigen is added and binds to the capture antibody.
- Detection antibody (often biotinylated) binds to a different epitope on the antigen, forming a “sandwich”.
- Enzyme conjugate (e.g., streptavidin‑HRP) binds to the detection antibody.
- Substrate (TMB) is added; HRP converts it to a colored product.
- Stop solution is added, and absorbance is read at 450 nm.
Direct, indirect, and competitive formats also exist, but the sandwich principle dominates for inflammatory cytokines and many other analytes. A high‑quality elisa kit manufacturer provides clear documentation of this workflow, including lot‑specific validation data.
Part 2: Core Quality Indicators and How to Test Them
When you receive a new batch of kits, or when comparing different suppliers, focus on these six essential parameters.
2.1 Sensitivity (Limit of Detection, LOD)
What it means: The lowest amount of analyte that can be reliably distinguished from zero.
How to verify:
Calculate LOD as the mean OD of 20 blank replicates + 3× their standard deviation. Then read this OD value off the standard curve to get the concentration in pg/mL.
Acceptable standard: A good quality ELISA test kit should have an LOD of ≤5 pg/mL for most cytokines. High‑sensitivity kits can reach 0.5–1 pg/mL.
2.2 Specificity (Cross‑reactivity)
What it means: The kit detects only the intended target, not structurally similar molecules or common interferents.
How to verify:
Test the kit against related molecules (e.g., for a human IL‑6 kit, test IL‑1β, IL‑8, TNF‑α) at high concentrations. Also test common matrix components (hemoglobin, bilirubin, lipids). Cross‑reactivity should be <1–5%.
Why it matters: A reputable elisa kit supplier will provide a cross‑reactivity table in the manual. If not, ask for one.
2.3 Precision (Intra‑assay and Inter‑assay CV)
What it means: The closeness of results when the same sample is measured repeatedly under the same (intra) or different (inter) conditions.
How to verify:
- Intra‑assay: Run the same sample in 5–10 replicates on one plate. Calculate the coefficient of variation (CV = SD / mean × 100%).
- Inter‑assay: Run the same sample on three different days, using different kit lots if possible.
Acceptable standard:
- Intra‑assay CV ≤10%
- Inter‑assay CV ≤15%
Values higher than these suggest poor batch consistency or manufacturing flaws.
2.4 Accuracy (Recovery)
What it means: The ability to measure the true concentration of the target.
How to verify:
Spike a known amount of standard into a real sample (e.g., serum) at low, medium, and high concentrations. Measure the spiked samples, then calculate recovery = (measured concentration – endogenous concentration) / spiked concentration × 100%.
Acceptable standard: Recovery between 80–120% is considered good. Values outside this range indicate matrix interference or antibody problems.
2.5 Stability
What it means: The kit retains its performance over time and under recommended storage conditions.
How to verify:
- Accelerated stability: Store the kit at 4°C and –20°C for periods corresponding to its shelf life, then test against fresh standards.
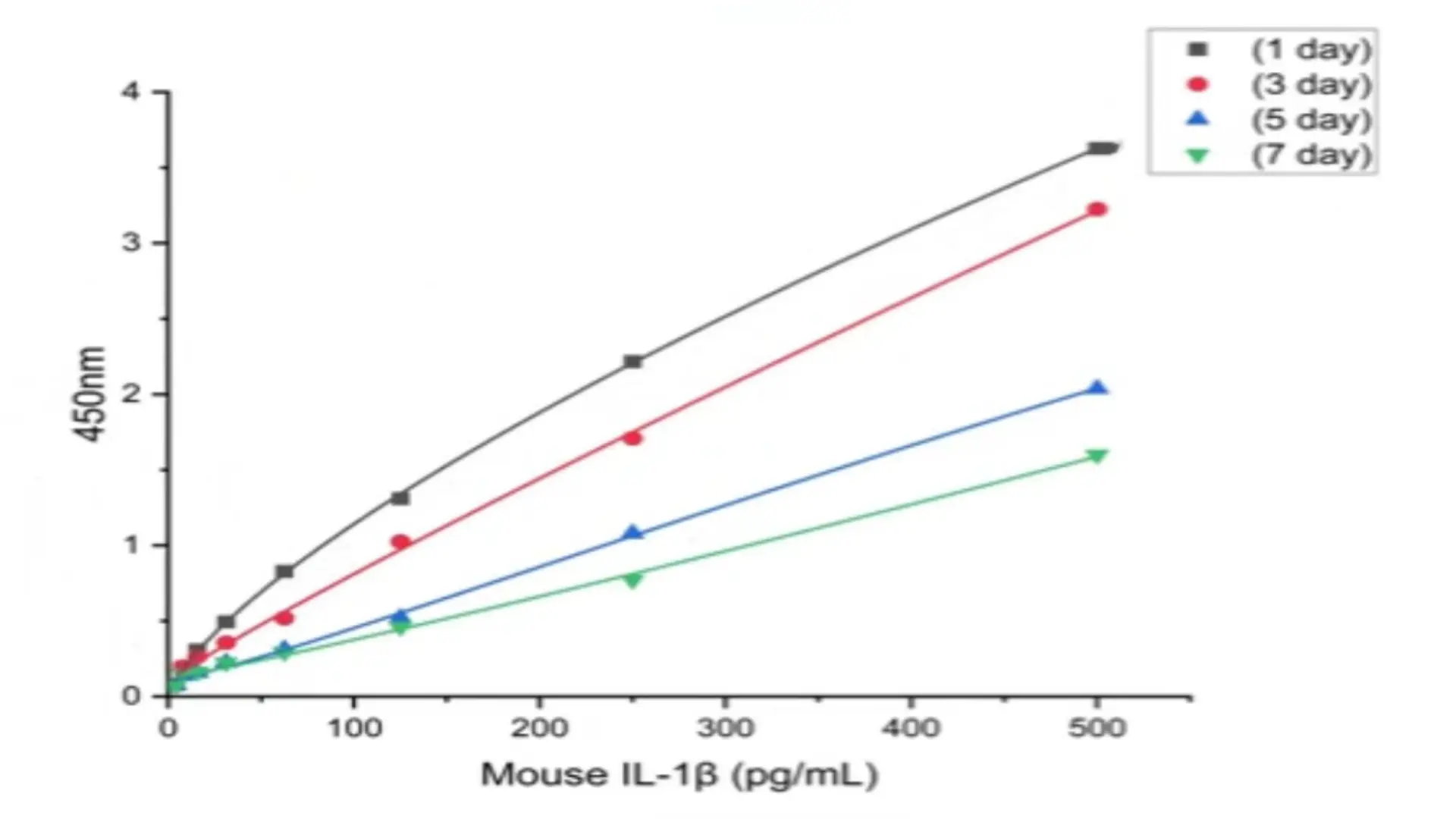
- Freeze‑thaw stability: Freeze and thaw the enzyme conjugate or detection antibody 3 times, then compare results to a never‑frozen control.
Acceptable standard: No significant change in OD or calculated concentration after 3 freeze‑thaw cycles. The kit should perform as advertised until the expiration date.
2.6 Linearity (Dilutional Linearity)
What it means: When a high‑concentration sample is serially diluted, the measured values should decrease proportionally.
How to verify:
Take a sample with high endogenous analyte (or a spiked sample). Perform 2‑fold or 4‑fold serial dilutions. Plot measured concentration against dilution factor. Perform linear regression; the R² value indicates linearity.
Acceptable standard: R² > 0.99 for the dilution series. Poor linearity suggests the kit’s dynamic range is too narrow or there are matrix effects.
Additional Quality Checks
- Standard curve quality: The correlation coefficient (R²) of the fitted curve (usually 4‑parameter logistic) should be ≥0.99.
- Appearance and packaging: Microplate wells should be free of scratches, cracks, or foreign material. Reagent labels must be legible, and liquids should show no turbidity or precipitation.
- Functional test: Run a known standard and a control sample. Compare the obtained concentration with the expected value.
Part 3: How to Determine If an ELISA Kit’s Precision Is Acceptable
Precision is the most frequently cited quality metric, but how do you decide if the numbers you calculate are truly “good enough”? Follow this structured approach.
3.1 Intra‑assay Precision Assessment
- Method: Run the same sample (preferably at low, medium, and high concentrations) in 3–5 replicates on a single plate.
- Calculation: Compute the mean, standard deviation (SD), and CV for each concentration.
- Acceptance criterion: CV ≤10% for all three levels. If CV exceeds 10%, check for pipetting errors, air bubbles, or uneven washing before blaming the kit.
3.2 Inter‑assay Precision Assessment
- Method: Test the same sample across 3 different runs, ideally on different days or with different kit lots. Use the same operator and protocol.
- Calculation: Calculate the CV of the results from all runs.
- Acceptance criterion: CV ≤15%. Higher values may indicate batch‑to‑batch variation or inconsistent storage conditions.
3.3 Standard Curve Stability
A precise kit produces a stable standard curve across runs. Plot the standard curves from different assays on the same graph; they should overlap closely. The R² of each curve should remain ≥0.99. If the curve shifts significantly (e.g., the top OD changes by >20%), the kit’s precision is questionable.
3.4 Quality Control Sample Validation
Use commercial or in‑house QC samples at three levels (low, medium, high). Run them in duplicate on every plate. Results should fall within the manufacturer’s expected range (often given as target value ±2SD). If QC samples fail, the kit’s precision is unacceptable for that run.
3.5 Compare with Industry Standards
Reference guidelines from CLSI (Clinical and Laboratory Standards Institute) or the FDA for assay validation. For research use, the criteria above (CV ≤10% intra, ≤15% inter) are widely accepted. For clinical diagnostic kits, stricter limits (Yanda BIO’s ELISA kit e.g., CV ≤8% intra, ≤10% inter) may apply.
3.6 Common Pitfalls That Skew Precision Metrics
Even a perfect kit can give poor precision if:
- Pipetting is inconsistent – use calibrated pipettes and fresh tips.
- Washing is incomplete – ensure wash buffer volume (300–400 µL) and correct number of cycles.
- Incubation times vary – use a timer and batch samples to avoid delays.
- Plates are not sealed – evaporation causes well‑to‑well variation.
If your precision test fails, repeat it with meticulous technique. If it still fails, contact your elisa kit manufacturer for a replacement lot or technical support.
Why Choose Yanda Bio as Your ELISA Kit Supplier?
At Yanda Bio, we are not just an elisa kit manufacturer – we are a partner committed to quality, transparency, and customer success. Our ELISA test kits are validated against all the metrics described above, and we provide full lot‑specific certificates of analysis so you can verify performance yourself.
- Thousands of targets – including inflammatory cytokines, growth factors, antibodies, and more, across human, mouse, rat, rabbit, and other species.
- Custom ELISA development – need a kit for a novel target? We will design and deliver a custom kit in as little as 5–7 business days.
- Uncompromising quality – intra‑assay CV ≤10%, inter‑assay CV ≤15%, recovery 80–120%, and R² ≥0.99.
- Affordable pricing – standard kits from just $120, with bulk discounts.
- Fast shipping – same‑day dispatch for orders before 3:30 PM (China time).
Final Thoughts
Verifying the quality of an ELISA kit does not require a PhD in immunology – just a few simple experiments and a willingness to check the numbers. By testing sensitivity, specificity, precision, accuracy, stability, and linearity, you can confidently choose a kit that will produce reproducible, trustworthy data. And when you need a reliable partner, remember that Yanda Bio delivers excellence in every box.